Incorporation of [2,3,4,5,6-2H5]Phenylalanine, [3,5-2H2]Tyrosine, and [2,4,5,6,7-2H5]Tryptophan into...
Applied Biochemistry and
Microbiology, Vol. 35, ffo. /. 1999, pp. 29-17. Translated from Prikladnayti Biokhimiya i
Mikrobialogiya, Vol. 35, No. 1,@ 1999, pp. 34-42. Original Russian Text Copyright © /999 hy Mosin, Skluclnev, Shvatz.
Incorporation of [2,3,4,5,6-2H5]Phenylalanine,
[3,5-2H2]Tyrosine,
and [2,4,5,6,7-2H5]Tryptophan
into the Bacteriorhodopsin Molecule of
Halobacterium halobium
O. V. Mosin*, D. A. Skladnev**, and V. I. Shvets*
*
Lotnonosov Moscow State Academy of Fine Chemical Technology, Moscow, 117571
Russia
** State
Center of Genetics and Selection of Industrial Microorganisms (GNU GENETICA),
Moscow, 113515 Russia
Received September 25, 1997
Abstract—Incorporation of
[2,3A5,6-2H5]phenylalanine, [3,5-2H2]tyrosine,
and [2,4,5,6,7-2H5]tryptophan into the
bacteriorhodopsin molecule followed by semipreparative isolation of
bacteriorhodopsin resulted in a yield of 8-10 mg per g bacterial biomass. This
method is based on the growth of the strain of halophilic bacteria Halobacterium
halobium on a synthetic medium containing 2H-labeled
aromatic ammo acids and fractionation of solubilized (in 0.5% sodium dodecyl
sulfate) protein by methanol, including purification of carotenoids. lip-ids, and
high-molecular-weight and low-molecular-weight compounds, as well as
gel-permeation chromatog-raphy on Sephadex G-200. Incorporation of 2H-labeled
amino acids was analyzed by electron impact mass spectrometry after
hydrolysis of the protein in 4 N Ba(OH)2 and separation in the form
of methyl esters of /V-DNS derivatives of amino aids by re versed-phase
high-performance liquid chromatography.
The
retinal-containing protein (a chromophore, pro-tonated aldimine of retinal
containing Lys-216 e-amino group) bacteriorhodopsin (BR), functioning as an
ATP-dependent translocase in cell membranes of halophilic bacteria Halobacterium
halobium was initially described by Oesterhelt [1]. In spite of the
fact that the structure
and functions of this protein were studied in detail, it is still a focus of
interest. This protein is used in practice
as a biological photochromic material because
of its high photosensitivity and resolution abilities [2]. Moreover, BR
is attractive as a model object for studies of the functional activity and
structural properties of membrane proteins
hi the composition of artificially designed energy-transforming
membranes.
The
introduction of isotopic labels into molecules of membrane proteins is appropriate for
studies of these proteins. Isotopic labels allow using the method of
high-sensitivity electron impact (El) mass spectrometry for further analysis of isotopic incorporation [3, 4]. Thus,
studies of BR labeled with the hydrogen isotope (deuterium) at residues of functionally important amino acids
(phenylalanine, tyrosine, and tryptophan) involved
in hydrophobic interaction of the protein polypeptide chain with the
lipid bilayer of the cell membrane are
important for practice [5, 6]. Raw 2H-labeled amino acids can
be readily synthesized in preparative
quantities by a reverse isotopic 1H-2H exchange in molecules of protonated amino acids, [2,3,4,5,6-2H5]phenylalanine
(in 85% 2H2SC>4 at50°C), [3,5-2H2]tyrosine (in 6 N 2H2SO4
at slight boiling), and [2,4,5,6,7-2H5]tryptophan
(in 75% [2H]trifluoroacetic acid
at 25°C) [7]. However, in spite of the rapid development of chemical
methods for obtaining 2H-labeled
aromatic amino
acids, the Russian industry of individual 2H-labeled
membrane proteins has not received wide acceptance.
This work was
designed to obtain sernipreparative quantities of 2H-labeled BR
for reconstruction of artificial membranes. Processes of incorporation of [2,3,4,5,6-2H5]phenylaIanine,
[3,5-2H2]tyrosine, and [2,4,5,6,7-2H5]tryptophan into the molecule of
bacteriorhodopsin followed with
further semipreparative isolation
were performed. The deuteration level was determined by means of El mass spectrometry performed after separation
of the protein hydrolysate in the form of
methyl esters of /V-DNS derivatives of amino aids by reverse-phase
high-performance liquid chromatography
(HPLC).
MATERIAL AND METHODS
Objects of
studies. The carotenoid-contain ing strain of extreme
halophilic bacteria Halobacterium halo-bium ET 1001
from the collection of cultures of microorganisms (Moscow State University) was
used. The strain was maintained on solid peptone medium (2% agar) containing
4.3 M NaCl.
Preparation
of growth media. DL-amino acids (Reanal, Hungary), adenosine
monophosphate (AMP) and uridine
monophosphate (UMP) (Sigma, USA), were
used. 5-[Dimethylamino]naphthalene-l-sulfonyl chloride (DNS chloride; Sigma, USA) and diaz-omethane obtained
from JV-nitroso-Af-methylurea (Merck, Germany) were applied for the synthesis
of amino acid derivatives. [2,3,4,5,6-2H5]Phenylalanine (90 at. % 2H), [3,5-2H2]tyrosine
(96 at. % 2H), and
[2,4,5,6,7-2H5]tryptophan
(98 at. % 2H) (methods for obtaining are described in [8, 9]) were
supplied by A.B. Pshenichnikova (Candidate of Chemical Sciences, Lomonosov Moscow State
Academy of Fine Chemical Technology).
2H-Labeled
BR. 2H-Labeled BR was
obtained on a synthetic medium, in which protonated ammo acids (phenylalanine,
tyrosine, and tryptophan) were replaced by their deuterium-containing analogues ([2,3,4,5,6-2H5]phenylalanine,
[3,5-2H2]tyrosine, and [2,4,5,6,7-2HJtryptophan). The medium contained 0.43
g/1 DL-alanine, 0.4 g/1 L-arginine,0.45 g/1 DL-aspartic acid, 0.05 g/1
L-cysteine, 1.3 g/1 L-glutamic acid, 0.06
g/1 L-glycine, 0.3 g/1 DL-histidine, 0.44 g/1 DL-isoleucine, 0.8 g/1 L-leucine, 0.85 g/1 L-lysine, 0.37 g/1
DL-methionine, 0.26 2/1 DL-phenylalanine, 0.05
g/1 L-proline, 0.61 g/1 DL-serine, 0.5 g/1 DL-thre-onine, 0.2 g/1 L-tyrosine, 0.5 g/1 DL-tryptophan,
1.0 g/1 DL-valine, nucleotides (0.1 g/1 AMP and 0.1 g/1 UMP), salts (250 g/I Nad, 20 g/1 MgSOa x 7H2O,
2 g/1 KC1, 0.5 g/1 NH4C1,
0.1 g/1 KNO3, 0.05 g/1 KH2PO4, 0.05 g/1 KoHPO4, 0.5 g/1 sodium citrate, 3 x 10
-4 g/1 MnSO4 x 2H2O, 0.065 g/1 CaCl2
- 6H2O, 4 x 10 -5 g/l ZnSO4 x 7H2O, 5 x 10 -5FeSO4
- 7H2O, and 5 x 10 -5 g/1 CuSO4 x 5H2O),
1 g/1 glycerin, and growth factors (1 x 10 -4 g/1 biotin, 1.5 x l0 -4 g/1 folic acid,
and 2 x 10 -5 g/1 vitamin
B!2).
Cultivation
of bacteria. The growth medium
was autoclaved for 30 min at 0.5 atm (pH was brought to 6.5-6.7
using 0.5 N KOH). The inoculum was grown in 750-ml Erlenmeyer's flasks (the medium
volume was 100 ml) on a 380-S orbital shaker
(Biorad, Hungary) at 35-37°C under conditions of intensive aeration and illumination (three LDS-40 lamps of 1.5 Ix each).
After 24 h, the inoculum (5-10%) was
transferred to the synthetic medium
and grown for five to six days (similarly to obtaining of the inoculum). All further manipulations for BR isolation were performed with the use
of a dimming lamp equipped with an ORZh-1
orange light filter.
Isolation
of the fraction of purple membranes (PM). The biomass (1 g) was
washed with distilled water and precipitated on a T-24 centrifuge (Carl
Zeiss, Germany) at 1500 g for 20 min. The precipitate was
suspended in 100 ml of distilled water and kept at 4°C. After 24 h,
the reaction mixture was centrifuged at 1500 g for 15 min. The precipitate was resuspended
in 20 ml of distilled water, disintegrated by sonication (2 kHz, three times
per 5 min) on a water bath containing ice (0°C), and centrifuged at 1500 g for 20 min. After washing with distilled water, the cellular homogenate was resuspended in 10 ml of buffer containing 125 mM NaCl, 20 mM MgCl2, and 4 mM Tris-HCl (pH
8.0). RNase (5 u,g, two-three units
of activity) was added. The mixture was incubated at 37°C. The same
buffer (10 ml) was added 2 h later. The
mixture obtained was kept at 4°C for 14-16 h. The water fraction was removed by
centrifugation at 1500 g for 20 min. The precipitate of
PMs was treated (five
times) with 7 ml of 50% ethanol at -5°C. The solvent was removed by
centrifugation at 1200 g and cooling for 15 min. The protein concentration was
measured on a DU-6 spectrophotometer (Beckman, USA) calculating the D280/D56S
ratio [10]. Regeneration of PMs was conducted as described in [11].
Isolation
of BR. The fraction of PMs (1 mg/ml) was
solubilized in 1 ml of 0.05% sodium dodecyl sulfate (SDS), kept at 37°C
for 7-9 h, and centrifuged at 1200 g for 15 min. The
precipitate was removed. Methanol (100 (ll) was added drop wise (three
times) to the supernatant at 0°C. The mixture was kept at -5°C for 14-15 h and then
centrifuged at 1200 g and cooling for 15 min. Fractionation was
performed three times with decreasing the concentration of SDS to 0.2% and
0.1%. Crystalline protein (8-10 mg) was washed with cold distilled water and centrifuged at
1200 g for 15 min.
Purification of BR. This procedure was performed by gel-permeation chromatography on a calibrated
column (150 x 10 mm). Sephadex G-200
(Pharmacia, USA) served as the
stationary phase (bed volume: 30-40 ml per g). The samples were taken manually.
The column was balanced with the
buffer solution containing 0.1% SDS
and 2.5 mM EDTA. The protein sample was
dissolved in 100 p.1 of the buffer solution and eluted with 0.09 M Tris-borate buffer (pH 8.5, / = 0.075) and 0.5 M NaCl at a flow rate of 10 ml/cm2
per h. Combined protein fractions
were subjected to lyo-philization.
Electrophoresis of
the protein. The procedure was
performed in 12.5% polyacrylamide gel (PAAG) containing 0.1% SDS.
The samples were prepared for elec-trophoresis by standard procedures (LKB protocol, Sweden).
Electrophoretic gel stained with Coomassie blue
R-250 was scanned on a CDS-200 laser densitom-eter (Beckman, USA) for quantitative analysis of the protein level.
Hydrolysis of BR. The
protein (4 mg) was placed into glass ampoules (10 x 50 mm in size), and 4 N Ba(OH)2 (5 ml) was added. The mixture
was kept at 110°C for 24 h. The
reaction mixture was suspended in 5 ml of hot distilled water and
neutralized with 2 N H2SO4
to pH 7.0. The sediment of BaSO4 was removed by centrifugation at 200 g for 10 min, and
the supernatant was evaporated in a
rotor evaporator at 40°C.
Synthesis
of N-DNS derivatives of amino acids. DNS chloride (25.6 mg) in 2 ml
of acetone was added gradually to 4 mg of
dry hydrolysate of BR in 1 ml of 2 M NaHCO3 (pH 9-10) under
conditions of constant mixing. The reaction mixture was kept at 40°C and mixing for 1 h, acidified with 2 N HCI to pH 3,
and extracted (three times) with 5 ml of ethyl acetate. The combined extract was washed with distilled water
to pH 7.0 and dried with anhydrous
Na2SO4. The solvent was removed at 10 mmHg.
Methyl
esters of N-DNS derivatives of amino acids. Wet
N-nitroso-.N'-methylurea (3 g) was added to 20 ml of 40% KOH in 40 ml of diethyl ether and
then mixed
on a water bath with ice for 15-20 min for
obtaining diazomethane. After the
completion of gas release, the ether layer was separated, washed with
distilled water to pH 7.0, dried with
anhydrous Na2SO4, and used for the treatment of /V-DNS
derivatives of amino acids.
Separation
of the mixture of methyl esters ofN-DNS derivatives of amino acids. This was
performed by the method of reverse-phase high-performance liquid chro-matography
on a Knauer liquid chromatograph (Germany) equipped with a Knauer pump, 2563
UV detector, and C-R 3A integrator (Shimadzy, Japan). The column of
250 x 10 mm in size was used. Separon C18 (Kova, Czech) served as
the stationary reverse phase. The diameter of granules was 12 urn. The
injection volume was 10 mkl. The following systems of solvents were used: (A)
acetonitrile and trifluoroacetic acid (at a volume ratio of 100 :
0.1-0.5) and (B) acetonitrile. Gradient elution processes were performed
at a rate of 1.5 ml/min for 5 min (from 0% to 20% B), 30 min (from 20%
to 100% B), 5 min (100% B), 2 min (from 100% to 0% B), and 10 min (0% B).
Mass
spectra. Mass spectra of methyl esters of N-DNS
derivatives of amino acids were obtained by the method of electron impact on an
MB-80 A instrument (Hitachi, Japan) at the energy of ionizing electrons of 70 eV,
accelerating potential of 8 kV, and a temperature of the cathode source
of 180-200°C. Scanning of the samples analyzed was performed at a resolution
of 7500
conditional units and a 10% image definition.
RESULTS AND DISCUSSION
Incorporation
of [2,3,4,5,6-2H5]phenylalanine, [3,5-2H2]tyrosine,
and [2,4,5,6,7-2H5]tryptophan into the
molecule of BR. The method of
incorporation of 2H-labeled amino acids into the molecule of BR
was selected because of the fact that this work was designed to reveal the
possibility for obtaining 2H-labeled preparations of the membrane
protein (in semipreparative amounts) for the reconstruction of
artificial membranes. [2,3,4,5,6-2H5]PhenyIalanine,
[3,5-2H2]ryrosine, and [2,4,5,6,7-2H5;]tryptophan
play important roles in hydrophobic interaction of the BR molecule with the lipid bilayer of the cell membrane. They are
stable to the 'H-2H
exchange in water medium under growth conditions.
Moreover, high-sensitivity El mass spec-trometry can be used for the analysis of their incorporation,
which was performed microbio logically by growing
the strain of halophilic bacteria Halobacte-rium halobium on
a synthetic medium containing 2H-labeled aromatic amino acids. Thus,
these compounds were selected as sources of
deuterium. Under the optimum growth conditions (exponential growth on a synthetic
medium with 4.3 M NaCl at 35-37°C and illumination), the cells
synthesized a purple pigment whose spectral
characteristics were identical to those of native BR. Figure 1 shows the dynamics of (2) bacterial growth on the medium containing -H-labeled
aromatic amino acids in relation to
(1) growth under control con-
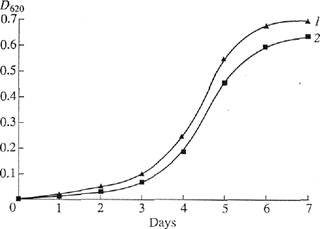
Fig. 1. The dynamics
of Che growth of Che strain//, halobium under various
experimental conditions: (/) protonated synthetic medium and
(2) synthetic medium with [2,3,4,5,6-2H5]phenylalanine, [3,5-2H2Jtyrosine,
and [2,4,5,6,7-2H5]tryptophan.
ditions. The growth of this strain on the medium containing 2H-Iabeled aromatic amino acids
was only slightly inhibited. This is
important for producing the raw 2H-labeled
biomass for further isolation of BR.
The main
stages of isolating 2H-labeled BR (Fig, 2) were the following:
production of 1 g of 2H-labeled bio-mass; isolation of
the fraction of PMs; removal of low-molecular-weight and high-molecular-weight admixtures,
cellular RNA, carotenoids, and lipids; fraction-ation of solubilized (in 0.05%
SDS) protein by metha-nol; and purification
on Sephadex G-200. Low-molecular-weight admixtures and the
intracellular contents were eliminated by
osmotic shock induced by distilled water
(after removing 4,3 M NaCl) followed by destruction of cell membranes by
ultrasound. The cellular homogenate was
then treated with RNase I (two-three units of activity) to induce the maximum
destruction of cellular RNA. The PM fraction obtained contained the complex of the desired protein with
Hpids and polysaccharides, as well
as admixtures of fixed carotenoids
and foreign proteins. Therefore, it was necessary to use special methods of protein fracdonation, which would not damage the native structure of
the protein native structure or cause its dissociation. This made the isolation of pure individual BR performed by
the use of special fine methods for removing carotenoids and lipids,
purification, and column chromatography more
difficult. Decarotenoidation was conducted by a repeated treatment of PMs with 50% ethanol at -5°C. Although it was a routine procedure, this stage
was necessary (despite of
considerable chromoprotein losses). The treatment was repeated no less than five times to obtain the absorption band of the PM suspension freed of carotenoids. Figure 3 shows (curves b, c) these
bands at various stages of
treatment in relation to (curve a) the band of
Growth of
Halobacterium halobium on synthetic medium containing [2,3,4,5,6-2H5]phenyIalanine,
[3,5-2H2]tyrosine and [2,4,5,6,7-2H5]tryptophan
|
Disintegration by
ultrasound
|
|
Water-soluble
products
of cellular content,
inorganic salts,
and other low-molecular-weight
compounds
|
 Distilled H2O
Distilled H2O
RNase I,
125 mM
NaCl, 20 mM MgCl,
4
mM Tris-HCl
Distilled H2O
Isolation of the biomass
Raw
biomass
________ t
Osmotic
shock
Culture
liquid
4.3 M
NaCl, and other
inorganic
salts
and
metabolites
50% ethanol
1.0.5%SDS-Na
2.
Methanol
-5°C
-5°C
PM
fraction
Decarotenoidation
±
Delipidation + BR
precipitation
—
Extract of carotenoids
_._
Residuals of cellular walls, lipids, and other high-molecular-weight
compounds
Crystalline
BR
t
Gel-permeation chromatography on Sephadex
G-200
4NBa(OH)7
UO°C,24h
1.
DNS
chloride, 2 M
NaHCO3, and ethyl acetate
2. jV-Nitroso-N-
methyl-_
urea, 40%
KOH
Purified BR ±
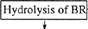
Mixture of free amino acids I
Modification into methyl esters
of /V-DNS derivatives of amino acids
Reverse-phase
HPLC
BaSO4
after neutralization with 2 M 2 M H2SO4
Individual
methyl esters of/V-DNS[2,3,4,5,6-2H5]phenylalanine
N-DNS-[3,5-2H2]tyrosine,
and N-DNS [2,4,5,6,7-2H5]tryptophan
El mass
spectrometry
Fig. 2. Experimentally designed method for
isolating H-labeled BR.
native BR. In this case, an 80-85%
efficiency of removing carotenoids was reached. The formation of the retinal-protein
complex induced a bathochromatic shift in the absorption band of PMs (Fig. 3). The
major band recorded at the maximum
absorption of 568 nm and induced by
the light isomerization of chromophore at
bonds positioned at C13=C14 or multiples of
this number was determined by the presence of trans-retinal residue of
retinal (BR568). The additional low-intensity band recorded at 412 nm characterized
the presence of a minor admixture of the M412
spectral form (produced in light)
containing the deprotonated aldirnine bond
 between the residue of trans-retinal
and the protein. The band recorded at 280 nm depended on the
absorption of
aromatic amino acids of the polypeptide chain of this protein (the D2%0/D56%
ratio was 1.5 : 1 for pure BR).
between the residue of trans-retinal
and the protein. The band recorded at 280 nm depended on the
absorption of
aromatic amino acids of the polypeptide chain of this protein (the D2%0/D56%
ratio was 1.5 : 1 for pure BR).
Fractionation
and careful chromatographic purification of the protein were the next necessary
stages. BR is a transmembrane protein with a
molecular weight of 26.7 kDa that penetrates the lipid bilayer in the form of seven a-helixes. Therefore, the use of ammonium
sul-fate and another traditional salt-eliminating agents is not appropriate. The protein must be transformed
into the soluble form by
solubilization in 0.5% SDS. The use
of this ionic detergent was dictated by the necessity of the most complete solubilization of the protein achieved
by combining delipidation and precipitation. In this case, BR solubilized in a
low-concentration solution of SDS retained
its helical cc-conformation [12].
Therefore, it was not necessary to use organic solvents such as
acetone, methanol, and chloroform for removing
lipids. Delipidation and precipitation of the protein were combined into the same stage. This noticeably
simplified fracdonation. The advantage of this method was that the desired
protein (in the complex with molecules of
lipids and detergent) was in the supernatant. Another
high-molecular-weight admixtures were in
the nonreacted precipitate, which was removed
by centrifugation. Fractionation of solubilized (in 0.5% SDS) protein and its further isolation in the crystalline
form were conducted using a gradual low-temperature
(-5°C) precipitation by methanol (three stages). The second and the third stages were performed by
decreasing the detergent concentration 2.5 and
5 times, respectively. The final stage of BR purification involved the
separation of the protein from low-molecular-weight
admixtures by gel-permeation chro-matography.
The fractions containing BR were passed two times through a column with dextran Sephadex G-200 balanced with 0.09 M Tris-borate buffer (pH 8.35)
containing 0.1% SDS and 2.5 mM
EDTA. The method designed for fractionation of the protein made it possible to obtain 8-10 mg of pure preparation of 2H-labeled
BR from 1 g of bacterial biomass. The homogeneity of BR complied with
the requirements on reconstruction of
membranes and was confirmed by electrophoresis in 12.5% PAAG with 0.1% SDS, regeneration of apomembranes
with trans-retinal, and reverse-phase HPLC
of methyl esters of N-DNS derivatives of amino aids. Low yield of BR was no barrier to further studies of isotopic
incorporation. However, it must be emphasized that considerable amounts of the raw biomass must be produced in
order to provide high yield of the protein.
Hydrolysis of BR. Conditions of hydrolysis of deuterium-containing protein were determined by the necessity of preventing the isotopic ('H-2H)
hydrogen-deuterium exchange in molecules of aromatic amino acids, as well as retaining tryptophan in the
protein hydrolysate. Two alternative
variants (acid and alkaline hydrolysis)
were considered. Acid hydrolysis of the
400 500 600 700
nm
Fig. 3. Absorption
bands (in 50% ethanol) at various stages of treatment: (a) native
BR, (b) PMs after intermediate treatment, and (c) P.Ms purified
of foreign admixtures. The band (/) corresponds to the spectral form of BR568.
The band (2) corresponds to the admixture of the M^ spectral form. The band (J)
characterizes the absorption of aromatic amino acids. The bands (4) and (5) correspond to
foreign caro-tenoids. Native BR was used as control.
protein performed
under standard conditions (6 N HC1 or 8 N H2SO4, 110°C,
24 h) is known to induce complete degradation of tryptophan and partial
degradation of serine, threonine, and several other amino acids in the protein [13]. These amino
acids do not play an important role in this
study. The modification of this method involving the addition of phenol [14],
thiogly-colic acid [15], and p-mercaptoethanol [16] into the reaction
medium allowed retaining tryptophan (to 80-85%).
7-ToIuenesulfonic acid with 0.2% 3-(2-aminoet-hyl)-indole, as well as 3
M 2-mercaptoethanesulfonic acid [18], are
the potent agents for retaining tryptophan (to 93% [17]). However, these methods are not suitable for
working the problem, because they have a noticeable weakness. Processes of the isotopic exchange (of a high
rate) of aromatic protons (deuterons) in molecules
of tryptophan, tyrosine, and histidine [19], as well as the exchange of
protons at C3 atom of aspartic acid and C4
atom of glutamic acid [20], proceed under conditions of acid hydrolysis. Thus,
the data on incorporation of
deuterium into the protein can not be derived from the hydrolysis performed even in deuterium-containing reagents (2HC1,2H2SO4,
and 2H2O).
Reactions of the
isotopic hydrogen exchange are nearly undetected (except for the proton (deuteron) at C2 atom of
histidine), and tryptophan is not degraded under conditions of alkaline
hydrolysis (4 N Ba(OH)2 or NaOH,
110°C, 24 h). Thus, this method of hydroly: sis was used in our
study. Simplification of the procedure for isolating the mixture of
free amino acids (due
Fig. 4. El mass spectrum of the mixture of
methyl esters of /V-DNS derivatives of amino acids of the BR hydrolysate.
Cultivation was performed on synthetic medium containing [2,3,4,5,6-
Hslphenylalanine, [3,5- H2]tyrosine, and [2,4,5,6,7-2H5]tryptophan.
Images
of molecular ions of arnino acids correspond to their derivatives (here and on
Fig. 5). Ordinate: relative intensity of the peak /)-
to neutralization with H2SO4)
was the cause of selection of 4 N Ba(OH)2 as a hydrolyzing agent.
Possible racemization of amino acids during alkaline hydrolysis did not affect
the results of further mass-spectrometry assay showing the deuteration level of
molecules of amino acids.
Study of
incorporation of [2,3,4,5,6-2H5]phenylala-nine, [3,5-2H2]tyrosine,
and [2,4,5,6,7-2H5]tryptophan into the
molecule ofBR. El mass spectrometry
following the modification of the mixture of free amino acids of the
protein hydrolysate into methyl esters of N-DNS derivatives of amino
acids was used for studies of incorporation of 2H-labeled
aromatic amino acids. Total El mass spectrum of the mixture of
methyl esters of N-DNS derivatives of 2H-labeled amino
acids was recorded to obtain reproducible data on the incorporation
of 2H-labeled aromatic amino acids. The deuteration level of
molecules was determined by calculating the difference between the values of
heavy peaks of molecular ions [M]+ enriched with deuterium of
derivatives of aromatic amino acids and their light unlabeled analogues.
Methyl esters of N-DNS derivatives of aromatic amino acids were separated by
reverse-phase HPLC, and El mass spectra of individual-amino acids were obtained. The El mass spectrum of the mixture
of methyl esters of N-DNS derivatives of amino acids (scanning at m/z 50-640, the base peak of m/z
527, 100%) was of the continuous type (Fig. 4). The peaks (in the range from 50 to 400 on the scale of mass
numbers) were represented by
fragments of metastable ions, low-molecular-weight admixtures, and
products of chemical modification of amino
acids. 2H-labeled aromatic
amino acids with mass numbers in the range
from 414 to 456 on the scale of mass numbers
were the mixtures of molecules containing various numbers of deuterium
atoms. Therefore, their molecular ions [M]+ were polymorphously
split (depending on the number of hydrogen atoms in the molecule) into individual clusters displaying static sets of m/z values.
Taking into account the effect of
isotopic polymorphism, the deuteration
level was determined from the most commonly encountered peak of the molecular ion [M]+ (which value was mathematically averaged by mass spectrometer)
in each cluster (Fig. 4).
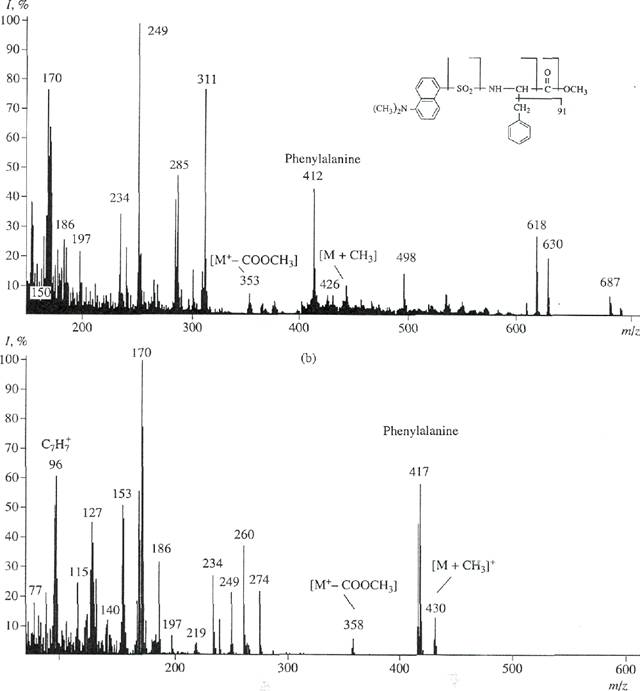
Phenylalanyne had a peak of a molecular
ion that corresponded to [M]+ and was 13% at m/z 417
(instead of [M]+ at m/z 412 for unlabeled phenylalanine;
peaks of unlabeled amino acids are not represented
here). Tyrosine had the peak of molecular ion that corresponded to [M]+ and was 15% at m/z 429
(instead of [M]+ at m/z 428). Tryptophan had a peak of a molecular ion that corresponded to [M]+
and was 11 % at m/z 456
(instead of [M]+ at m/z 451). Levels of deuteration
corresponding to the increase in molecular weights
were one (for tyrosine) and five (for phenylalanine and tryptophan) atoms of deuterium. These results showing deuteration levels of phenylalanine,
tyrosine, and tryptophan are in agreement with data on the deuteration levels
of initial amino acids. This indicates a sufficiently high potency of incorporation of 2H-labeled aromatic
amino acids into the protein molecule. Thus, incorporation
of 2H-labeled amino acids into the BR molecule was of a specific type. Deuterium was detected in all residues of aromatic amino acids.
However, it should be stressed that
there were [M]+ peaks of protonated
and semideuterated analogues of phenylalanine with [M]+ at m/z
414 (20%), 415 (18%), and 416
|
|
Fig, 5. El mass spectrum of the mixture of
methyl esters of N-DNS phenylalanine under various experimental conditions: (a)
unla-beled methyl ester of N-DNS phenylalanine and (b) methyl ester of
/V-DNS [2,3,4,5,6-2H5] phenylalanine isolated by
reverse-phase HPLC.
(11%); tyrosine with [M]+ at m/z428 (12%); and
tryp-tophan with [M]+ at m/z 455 and 457 (9%) displaying various contributions to the deuteration level of
molecules. This suggests that small part of minor pathways of their biosynthesis de novo leading to
the dilution of a deuterium label was retained. The presence of these peaks probably depended on conditions of
biosynthetic
incorporation of 2H-labeled aromatic amino acids into the protein molecule.
The
analysis of scan El mass spectrum showed that peaks of molecular
ions [M]+ of methyl esters of N-DNS derivatives of aromatic amino acids
had low intensities and were polymorphously
split. Therefore,
their molecular
enrichment ranges were considerably
widened. Moreover, mass spectra of the
mixture components were additive. Therefore, these mixtures can be analyzed only in the case of
the presence of spectra of various components recorded under the same conditions. These calculations involve solution of the
system of n equations in n unknowns for the mixture
containing n components. For the
components, whose concentrations
are more than 10 mol %, the validity and repro-ducibility of the analysis results can be ±0.5 mol % at a confidence probability of 90%. Therefore,
chromato-graphical isolation of individual
derivatives of 2H-labeled amino acids from the protein
hydrolysate is necessary for a obtaining a
reproducible result. Reverse-phase HPLC on octadecylsilane silica gel, Separon C18 (whose potency was
confirmed by separation of methyl
esters of //-DNS derivatives of 2H-labeled amino acids of another microbial objects, e.g., methylotrophic bacteria and microalgae [21]), was used. This method was adapted to conditions of
chro-rnatographical separation of a
mixture of methyl esters of DNS
derivatives of amino acids of the BR hydrolysate.
Optimization of eluant ratios, the gradient type, and the rate of elution from the column were performed. The maximum separation was observed after
gradient elution with a mixture of solvents containing acetonitrile and trifluoroacetic acid (at a
volume ratio of 100 : 0.1-0.5). In
this case, tryptophan and a hardly degraded
pare of phenylalanine/tyrosine were successfully separated. Degrees of
chromatographical purities of
isolated methyl esters of N-DNS [2,3,4,5,6-2H5]phe-nylalanine, N-DNS [3,5-2H2]tyrosine,
and N-DNS [2,4,5,6,7-2H5]tryptophan
were 97%, 96%, and 98%, respectively.
The yield was 97-85%. Figure 5b confirms
the result obtained. This figure shows the El mass spectrum of methyl ester of N-DNS [2,3,4,5,6-2H5]phe-nylalanine
isolated by reverse-phase HPLC (scanning at m/z
70-600; the base peak at m/z 170; 100%). The mass spectrum is
represented in relation to unlabeled methyl ester of//-DNS phenylalanine
(scanning at m/z 150-700; the base
peak at m/z 250; 100%) (Fig. 5a). The peak of a heavy molecular ion of methyl ester of N-DNS phenylalanine ([M]+, 59% at m/z
417; instead of [M]+, 44% at m/z 412 for unlabeled derivative of
phenylalanine) and the additional peak of the benzyl fragment of phenylalanine, C7H7
(61% at mlz 96; instead of
55% at mlz 91 for control; data not shown), confirm the presence of
deuterium in phenylalanine. The peaks of
secondary fragments of various intensities with m/z 249, 234, and 170 correspond to products of
secondary degradation of the dansyl
residue to N-dimethylaminon-aphthalene.
The low-intensity peak of [M+-COOCH3] (7%) at m/z 358 (m/z 353, 10%,
control) represents the detachment of the carboxymethyl group from
methyl ester of N-DNS phenylalanine. The
peak of [M + CH3]+ (15%) at m/z 430 (m/z 426, 8%, control) represents the
additional methylation at a-amino group of phenylalanine. The difference between molecular weights of
light and heavy peaks of [M]+of
methyl ester of N-DNS phenylalanine is five units. This is in
agreement with the earlier obtained result and the data on the level of
deutera-tion of initial [2,3,4,5,6-2H5]phenylalanine added
into the growth medium.
Thus, these data
indicate a high efficiency of incorporation of 2H-labeled aromatic amino acids into
the BR molecule. Completely deuterated
protein preparations for reconstruction (into 2H2O)
of functionally active systems of membrane
proteins with purified 2H-labeled
lipids and other deuterated biologically active compounds are proposed to be obtained using the method elaborated. In the future, these studies
will provide the means for solving
the problem of functioning of 2H-Iabeled
BR in the composition of artificially constructed membranes under conditions of deuterium-saturated medium.
ACKNOWLEDGMENTS
This work was
supported by grant no. 1B-22-866 ("High chemical technologies"). We
are grateful to Dr.
B.M. Polanuer (GNU GENETICA) for careful attention
and helpful remarks in discussions of the results.
REFERENCES
1.
Oesterhelt,
D. and Stoeckenius, W., Nature (London),
1971, vol. 233, no 89, pp. 149-160.
2.
Spudich, J.L., Ann. Rev.
Biophys. Chem, 1988, vol. 17,
no. 12, pp. 193-215.
3.
Karnaukhova,
E.N., Niessen, W.M.A., andTjaden, U.R.,
Anal
Biochem., 1989,
vol. 181, no. 3, pp. 271-275.
4.
Mosin,
O.V., Skladnev, D.A., Egorova, T.A., and
Shvets, V.I., Bioorg.
Khim., 1996, vol. 22, nos. 10-11,
pp. 856-869.
5.
Hardy,
J.R, Knight, A.E.W., Ghiggino, K.R, Smith, T.A.,
and Rogers,
P.J., Photochem. Photobiol, 1984, vol. 39,
no. 1, pp. 81-88.
6.
Rosenbach,
V., Goldberg, R., Gilon, C., and Ottolenghi, M.,
Photochem.
Photobiol, 1982,
vol. 36, no. 6, pp. 197-
201.
7.
Mosin,
O.V., Skladnev, D.A., Egorova, T.A., and
Shvets, V.I., Biotechnologiya, 1996,
no. 10, pp. 24-40.
8.
Griffiths,
D.V., Feeney, J., Roberts, G.C., and Burgen, A.S.,
Biochim.
Biophys. Acta, 1976, vol. 446, no. 4, pp. 479-585.
9. Matthews, H.R.,
Matthews, K.S., and Opella, S.J., Bio
chim. Biophys. Acta, 1977, vol. 497, no. 23, pp. 1-13.
10. Oesterhelt,
D. and Hess, B., Eur. J. Biochem., 1973,
vol.
37, no. 1, pp. 316-326.
11.
Tokunada,
F. and Ebrey, T, Biochemistry, 1978, vol. 17,
no. 10, pp.
1915-1922.
12.
Pervushin,
K.V. and Arsen'ev, A.S., Bioorg. Khim.,
1995, vol. 21, no. 10, pp.
83-111.
13.
Zvonkova,
E.N., Zotchik, N.V., Filippovich, E.I., Mitro-
fanova, T.K.,
Myagkova, G.I., and Serebrennikova, G.A.,
Khimiya biologicheski aktivnykh
prirodnykh soedinenii
(Chemistry of Biologically Active
Natural Compounds), Moscow:
Khirniya, 1970, pp. 65-68.
14.
Muromoto,
K., Sunahara, S., and Kamiya, H., Agric.
Biol.
Chem., 1987,
vol. 51, no. 6, pp. 1607-1616.
15.
Matsubara,
H. and Sasaki, R.M., Biochim. Biophys. Res.
Com., 1969, vol. 35, no. 10, pp. 175-177.
16.
Ng,
L.T., Pascaud, A., and Pascaud, M., Anal. Biochem.,
1987, vol. 167, no. 2, pp.
47-52.
17.
Liu,
T.Y. and Chang, Y.H..J.Biol. Chem., 1971, vol. 246,
no. 2, pp.
2842-2848.
19.
Pshenichnikova,
A.B., Karnaukhova, E.N., Zvonko
va, E.N., and
Shvets, V.I., Bioorgan. khimiya, 1995,
vol. 21, no.
3, pp. 163-178.
20.
Cohen,
J.S. and Putter, I., Biochim. Biophys. Acta, 1970,
vol. 222, no.
1, pp. 515-520.
2E. Egorova, T.A., Mosin, O.V.,
Eremin, S.V., Karnaukhova, E.N., Zvonkova, E.N., and Shvets, V.I., Bio-technologiya, 1993, no. 8, pp. 21-25.